The U.S. Food and Drug Administration approved a new drug on Friday that reduces the complications associated with sickle cell disease — the first drug approved for the blood disorder in more than 20 years. The drug, called Endari, consists of L-glutamine, which is an amino acid, and is approved for sickle cell patients five years and older.
“I am hoping we are finally seeing channels opening, and that this will be the first of many new drugs to hit the market for sickle cell disease ,” said Dr. Alexis A. Thompson, head of the Hematology Section and Director of the Comprehensive Thalassemia Program at the Ann and Robert H. Lurie Children’s Hospital of Chicago.
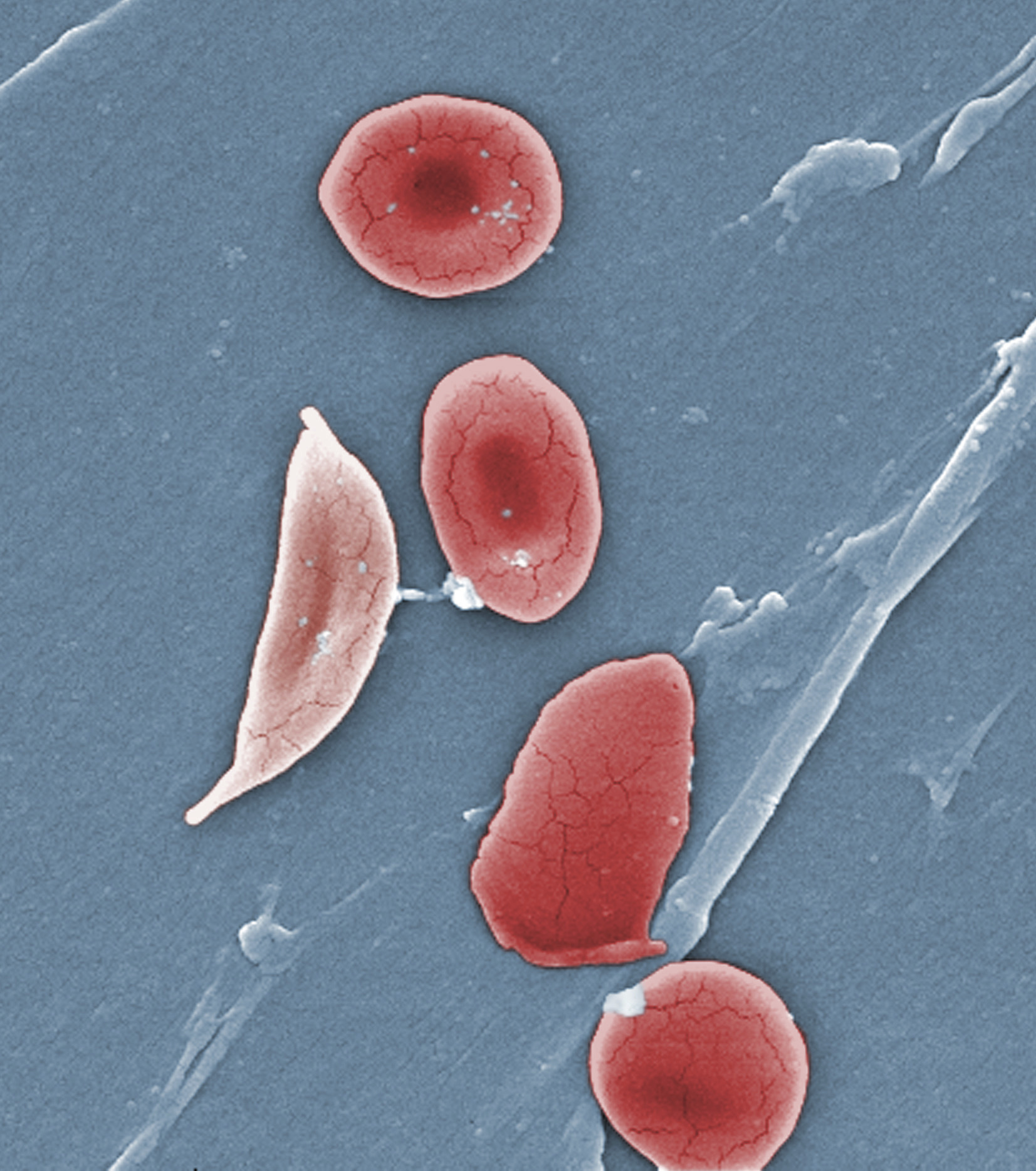
Sickle cell disease, which predominately affects African-Americans, Latinos and other minority groups, is an inherited lifelong disorder in which red blood cells that are normally round and disc-shaped are instead crescent or sickle-shaped due to abnormal hemoglobin. These sickle-shaped cells are not flexible, and they can stick to the walls of blood vessels, causing blockages that stops the flow of blood to organs or tissues. This is the hallmark of the painful, sometimes debilitating, sickle cell pain crises which often require an Emergency Department visit or hospitalization. The crises can occur without warning or can be triggered by stressors such as cold weather, dehydration or pregnancy.